As vaccine technology advances, more specialized immunogens that offer better protection against new or evolving diseases are being created, as evidenced by the diversity of SARS-COV-2 vaccines. But these vaccines often lack the inherent triggering power of live or inactivated pathogens.
Adjuvants stimulate stronger and longer-lasting immune reactions, improving vaccine effectiveness. They are especially helpful for elderly, immunocompromised, and pediatric patients who do not generate effective responses to vaccines.
Until recently, few adjuvants were available for clinical trials and use in humans. However, with changes in the intellectual property landscape, there is a renewed opportunity for companies to specialize in adjuvant development and supply to vaccine developers.
This is where EVAX adjuvants come in: we offer the unique opportunity to be your adjuvant partner during your initial pre-clinical work and supplier for your clinical trials and beyond. It is never too early to consider your supply chain. Using the same ingredients and formulations throughout your work can save time and incredible costs when it is time to move into the clinic.
EVAX Adjuvanted Vaccines Have Demonstrated Bioequivalence
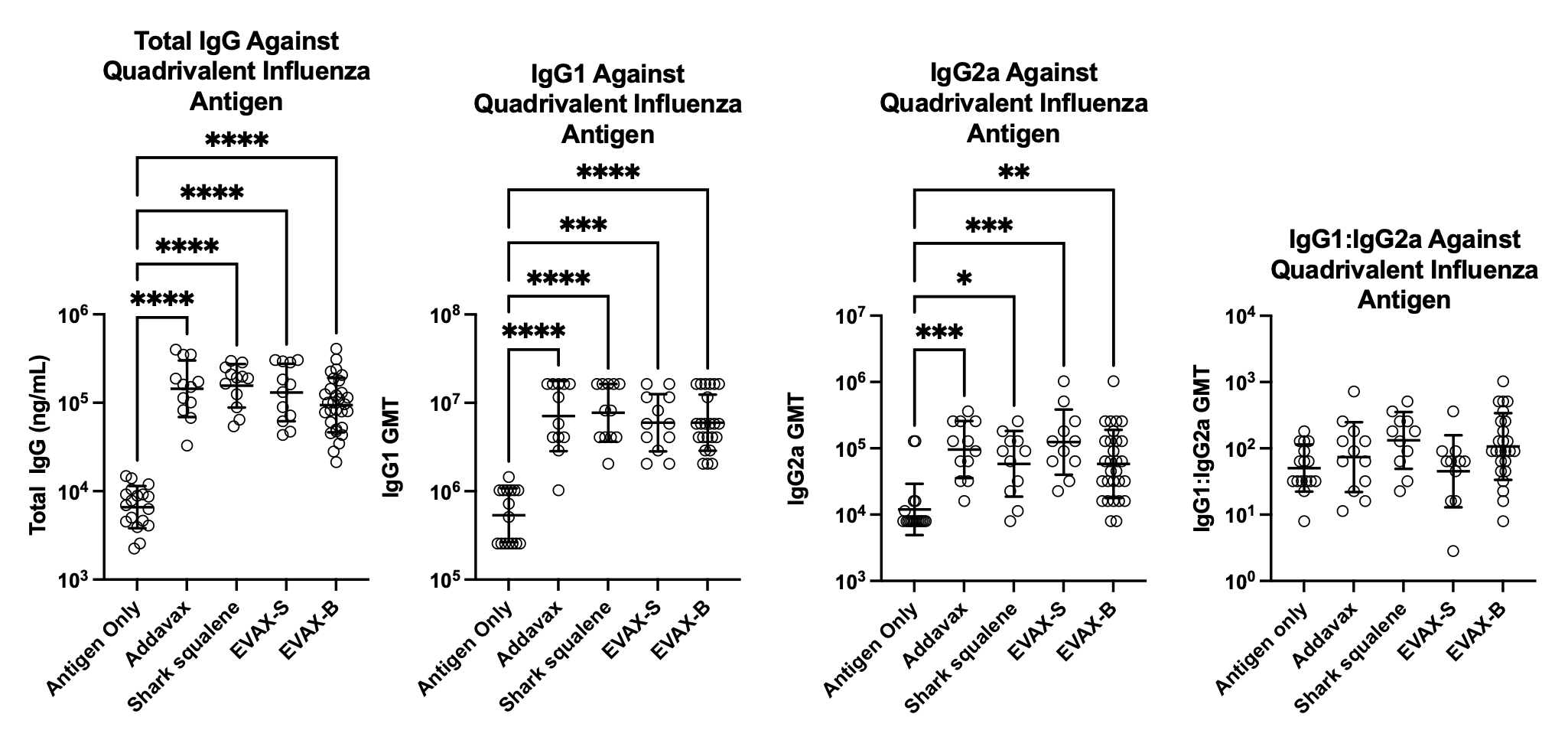
EVAX adjuvants have been tested with inactivated (split-viron) influenza, inactivated influenza viral particles, and recombinant protein SARS-CoV-2 antigens. In each of these, it showed immunoglobulin and hemagglutination inhibition, and/or neutralizing antibody titers equivalent to other commercial emulsion adjuvants.